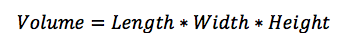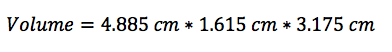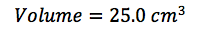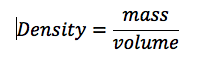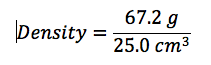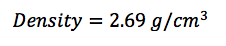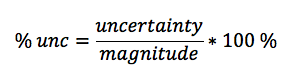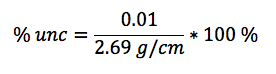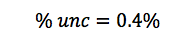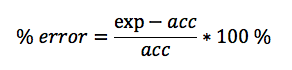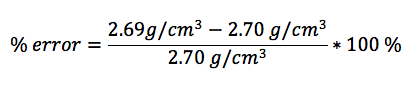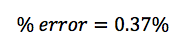Density Lab
Saige Miller
9-4-15
Purpose: The purpose of this lab is to determine the density of several objects, using various tools to make precise measurements in order to calculate the density.
Theory: The density of a given object can be determined by the equation P=M/V, or density equals mass divided by volume.
Experimental Technique:
Conclusion
In this lab, we calculated the density of several objects. Once calculated, it was discovered that the calculated density of each object varied from the accepted density of the same object. This is due mainly to errors in the lab. These errors include human and instrumental error. For example, while measuring the mass of aluminum, the scale only reads to the tenths place. If it were possible to calculate the mass of the aluminum for precisely, the density may have been closer to the accepted value. Another source of error would simply be human error. Human error occurs when data is interpreted from the instruments used. A vernier caliber was used to measure the length, width, and height of each object. While taking each measurement, it is possible to misjudge the correct number, throwing off the values of the dimensions of the object. By eliminating or reducing each source of error, the errors in density will become more accurate to the accepted value.